Introduction
Indigenous communities and isolated rural populations face a range of health challenges shaped by longstanding social, environmental, and structural conditions. Both groups experience a considerable burden of chronic diseases, infectious illnesses, undernutrition, and mental health conditions, compounded by limited opportunities for preventive care and health promotion1-3. These health patterns are linked to shared circumstances, including lower household income, reduced access to education, and limited availability of essential public services4,5. Geographic isolation further restricts access to timely health care and reduces engagement with routine services.
Although they share many of these constraints, Indigenous Peoples also encounter challenges that are rooted in historical and cultural contexts. The continuing effects of colonisation, land dispossession, cultural disruption, and experiences of discrimination within health systems contribute to longstanding inequities in health and wellbeing1,6-8. These factors influence trust in mainstream services, shape patterns of care-seeking, and deepen barriers to sustained engagement with formal health care2,9.
These combined circumstances have important implications for maternal health. Maternal healthcare services have a central role in improving maternal and neonatal outcomes, yet Indigenous and rural populations often face heightened barriers to accessing quality care. Obstacles such as geographic remoteness, cultural differences, economic constraints, and historical mistrust of healthcare systems can limit timely antenatal attendance and safe delivery options10,11. As a result, these communities continue to exhibit poorer maternal health indicators including higher rates of maternal mortality, preterm birth, and delivery complications compared with more advantaged populations12-14. In response, health authorities have introduced maternal healthcare programs as part of priority healthcare systems tailored to the needs of Indigenous and rural communities, seeking to improve access, continuity of care, and cultural safety.
These interventions aim to enhance prenatal care, ensure safe delivery practices, and provide comprehensive postnatal support, all while being culturally sensitive and accessible. For instance, culturally tailored prenatal programs have shown promise in increasing the utilisation of antenatal care, improving birth outcomes, and reducing complications15,16. Additionally, community-based birthing centres, staffed with skilled midwives familiar with Indigenous cultural practices, have effectively ensured safe delivery practices by providing accessible and culturally respectful environments for childbirth17,18. Postnatally, home-visiting programs that offer breastfeeding support, newborn health monitoring, and maternal mental health services have been instrumental in delivering continuous, culturally appropriate support during the critical postpartum period19.
Despite the documented clinical benefits of these programs, there is a notable lack of comprehensive economic evaluations. Conducting such evaluations is essential to determine how resources can be allocated efficiently and whether these programs are financially sustainable over time. Economic evaluation is a comparative analysis of the costs and outcomes of two or more health interventions, carried out to determine which option provides better value for the resources used.
In the literature, types of economic evaluation include cost-effectiveness analysis (CEA), cost–utility analysis (CUA), cost–benefit analysis (CBA), and cost–consequence analysis (CCA). CEA compares interventions based on natural units of outcome, such as cases detected or complications avoided. CUA incorporates broader measures of health by translating outcomes into utility-based metrics, most commonly the quality-adjusted life year (QALY). CBA expresses both costs and outcomes in monetary terms to determine whether the net benefits of an intervention justify its investment. CCA presents costs alongside a range of outcomes without aggregating them into a single summary measure, allowing decision-makers to weigh the various consequences according to their priorities.
QALYs provide a combined measure of survival and health-related quality of life, enabling comparisons across diverse health interventions. In contrast, disability-adjusted life years (DALYs) reflect the burden of disease by quantifying years of healthy life lost due to both premature mortality and time lived in less-than-ideal health. Together, these measures support more consistent and transparent comparisons of interventions when conducting CUA or burden-of-disease analysis.
Policymakers and healthcare providers need robust economic evidence to make informed decisions that balance quality, accessibility, and financial feasibility. Without such data, there is a risk of either underfunding effective programs or overinvesting in interventions that may not deliver proportional economic benefits.
Therefore, this systematic review aims to evaluate the current evidence on the economic effectiveness of maternal healthcare services for Indigenous and rural populations. By assessing economic evaluation studies, this review seeks to identify gaps in the literature, provide insights into the most efficient use of resources, and offer recommendations for future research and policy development in maternal health services tailored to these communities.
Methods
This systematic review adhered to PRISMA (Preferred Reporting Items for Systematic reviews and Meta-Analyses) 2020 guidelines, with defined protocols for study selection, data extraction, and quality appraisal.
Eligibility criteria
Included studies were full economic evaluations of maternal health interventions targeting Indigenous and/or rural populations. Eligible populations included those identified as Indigenous, tribal, Aboriginal, First Nations, or rural. Mixed-population studies were included if results were disaggregated or clearly relevant.
Eligible interventions spanned clinical, structural, or financial strategies during antenatal, intrapartum, or postnatal periods. These included direct health services (eg antenatal care, skilled birth attendance), outreach (eg home visits, mobile clinics), and culturally adapted care.
Studies had to compare both costs and outcomes between alternatives. CCA analyses were accepted if cost and health outcome data were clearly reported. Cost-only analyses, return on investment, and budget impact studies were excluded.
Primary outcomes were economic metrics (eg incremental cost-effectiveness ratios (ICERs), cost per QALY / disability-adjusted life year (DALY)). Secondary outcomes included service indicators like antenatal coverage, maternal/neonatal morbidity, and skilled birth attendance.
Only studies published between 1 January 2004 and 30 April 2025 were included. Acceptable study designs included randomised controlled trials, quasi-experiments, observational studies, and model-based economic evaluations.
Search strategy
The following databases were searched: PubMed, Cochrane Library, International Health Technology Assessment (HTA) Database, ProQuest, Cost-Effectiveness Analysis Registry, and the Centre for Reviews and Dissemination. Searches spanned 1 January 2004 to 30 April 2025, using MeSH and free-text terms (Appendix I). Grey literature included unpublished reports and non-government organisation publications. Duplicates were removed using Zotero organisational software v6.0.36 (Digital Scholar; https://www.zotero.org).
Screening and selection
Two authors (AAAN and LSS) screened titles, abstracts, and full text independently. Disagreements were resolved by discussion or by a third author (KI). Reasons for exclusion were recorded and illustrated in the PRISMA diagram.
Data extraction and quality assessment
Two authors (AAAN and KI) extracted data independently using a standardised form to capture study characteristics, population, intervention details, evaluation type, and outcomes. No automation tools were used. Discrepancies were resolved by consensus. If required information was missing or unclear in the published reports, we recorded it as ‘not available’. No imputations were made. Unreported variables were excluded from synthesis and flagged in quality appraisal.
Costs were converted to 2024 US dollars (1 USD = 1.45 AUD) using the C-CEMG-EPPI cost converter (EPPI Centre; https://eppi.ioe.ac.uk/costconversion) with purchasing power parity adjustments20. For primary outcomes, effect measures included ICERs expressed as cost per QALY or DALY gained. Secondary outcomes were summarised descriptively; no pooled effect measures were calculated due to heterogeneity across studies.
Quality was assessed using the CHEC (Consensus Health Economic Criteria)-Extended checklist21,22. This 20-item checklist is applicable to both model-based and trial-based economic evaluations. Each item was scored as 1 for a positive response and 0 for a negative one. The total score was calculated by summing the individual item scores and expressing it as a percentage (total score ÷ 20 × 100%). Based on this percentage, studies were categorised into four quality levels: low (≤50%), moderate (51–75%), good (76–95%), and excellent (>95%). A higher percentage reflects better methodological quality. For the risk of bias, we assessed the included studies using the Bias in Economic Evaluation (ECOBIAS) checklist23. This tool comprises two main components. The first addresses overall bias across 11 domains: narrow perspective, inappropriate comparator, omission of relevant costs, sporadic data collection, flawed valuation methods, ordinal ICERs, double-counting, improper discounting, limited sensitivity analysis, funding bias, and selective reporting. The second component focuses on bias in model-based economic evaluations, covering three subdomains: model structure (four items), data inputs (six items), and internal consistency (one item). Each criterion was rated as ‘yes’, ‘no’, ‘partly’, ‘unclear’, or ‘not applicable’, with ‘yes’ indicating high risk of bias and ‘no’ indicating low risk of bias.
Data synthesis and analysis
A narrative synthesis approach was used due to methodological heterogeneity. Findings are presented in structured summary tables organised by intervention type, population focus and outcome category, and were synthesised descriptively. No subgroup analysis or meta-regression was performed. No statistical methods were used to assess reporting bias. During synthesis, variations in national healthcare systems, financing models, Indigenous health policies, resource availability, and geographic context were considered when interpreting cost-effectiveness findings across countries.
Ethics approval
This study is a systematic review of previously published data. No primary data collection or involvement of human participants occurred. Therefore, institutional review board approval and informed consent were not required.
Results
Study selection and characteristics
From 1095 records and four manually added studies, 1000 were screened. After full-text review, 42 studies met the inclusion criteria, yielding 50 economic evaluations (Fig1). These included 30 CEAs, 15 CUAs, and 5 other formats (eg CCA). Evaluations spanned 25 countries, with India (n=7) and Uganda (n=4) most represented. Summary results of all included evaluations are presented in Appendix II24-65.
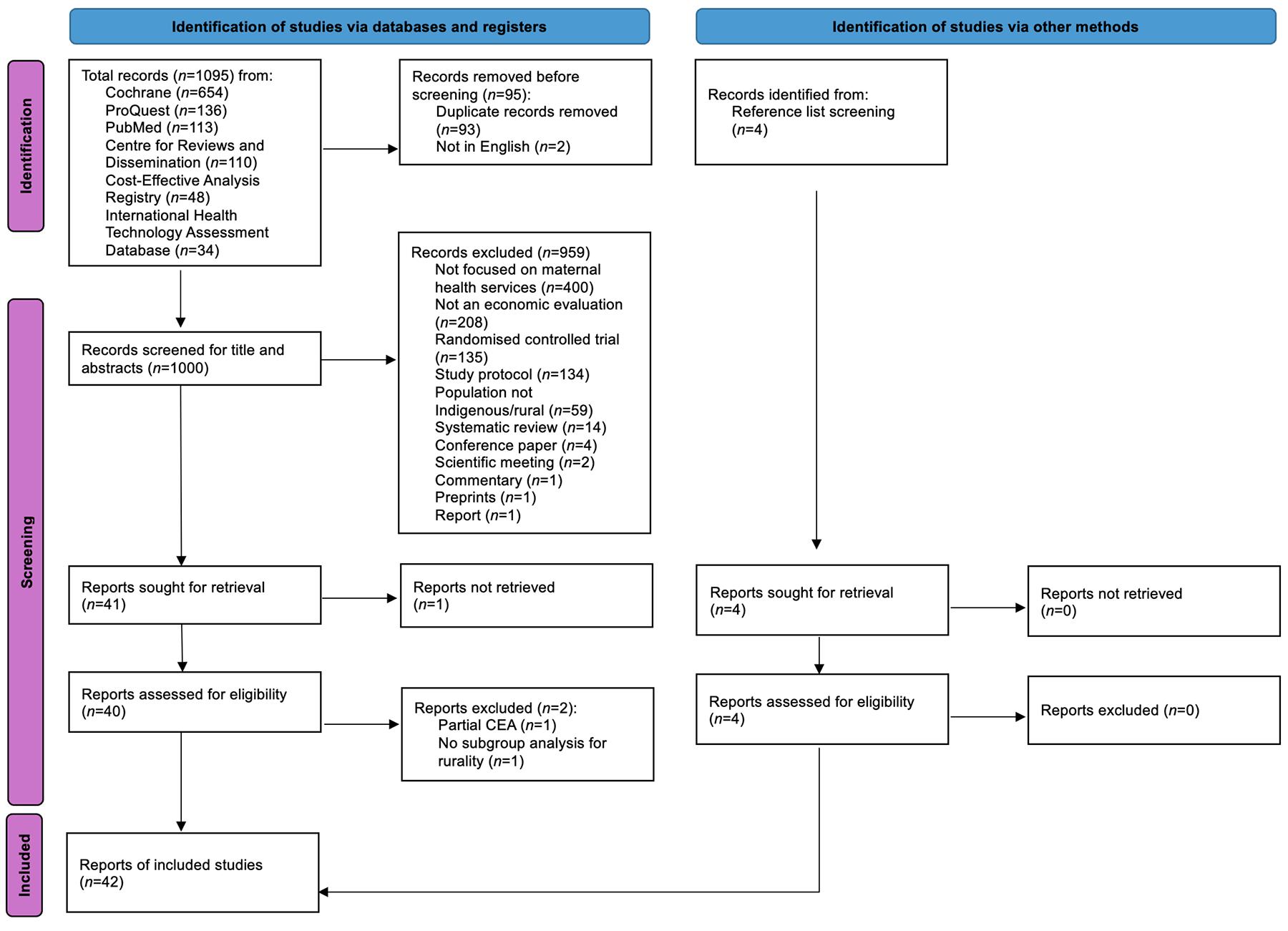 Figure 1: PRISMA 2020 flow diagram for new systematic reviews, which included searches of databases, registers and other sources.
Figure 1: PRISMA 2020 flow diagram for new systematic reviews, which included searches of databases, registers and other sources.
Risk of bias
The ECOBIAS checklist showed that most studies (20/22 criteria) had a low risk of bias (Appendix III24-65). Common issues included narrow costing perspective and limited sensitivity analyses. Most studies selected comparators well and reported transparently.
Study quality
Over half of the studies scored ≥95% on the CHEC-Extended checklist (Appendix IV24-65). Lower-rated studies lacked clarity on time horizons, modelling methods, discounting, or uncertainty analysis.
Thematic analysis by intervention type
Thematic analysis of the included studies revealed five dominant intervention categories: community-based care (n=15), clinical or diagnostic innovations (n=9), system strengthening (n=8), digital and mobile health tools (n=6), and financial incentives (n=6).
Community-based interventions
Community-based interventions were the most commonly evaluated, often engaging community health workers (CHWs)30,34-36,45,49,52,54, women’s groups38,41,45,47,58, midwives29,32,49, or traditional birth attendants43 or broader community mobilisation actors51. These programs aimed to increase care-seeking behaviours, improve cultural safety, and deliver care in familiar, accessible settings. Studies showed strong cost-effectiveness across diverse contexts. For example, participatory learning and action groups in India and Nepal were associated with reduced neonatal mortality at a cost per DALY averted ranging from US$49 to US$10445,58. In Australia, the Birthing in Our Community model, which provided culturally safe midwifery care for First Nations women, was cost-saving and effective, reporting a US$3909 saving per mother–baby pair32. CHW home-visit programs in Bolivia, Mali, and Vietnam also demonstrated favourable economic outcomes. For example, the cost per cognitive point gained in a child ranged from US$7 to US$20, as measured using the Bayley Scales of Infant and Toddler Development, third edition (Bayley-III)35,36. The cost per mother–child pair reached US$38130, while the ICER per additional antenatal care (ANC) contact compared to standard care was reported as low as US$2.0252.
A study in Sierra Leone in which the researchers ‘rebranded’ traditional birth attendants as maternal and newborn health promoters found that combining health promotion with a social enterprise model yielded greater health impact and cost-effectiveness (US$1963 per life year saved) than health promotion alone43.
Similarly, in rural Burkina Faso, a demand-side intervention involving district-wide community mobilisation and behavioural change communication was positively correlated with a sharp increase in institutional deliveries. The narrow incremental cost per additional delivery attributed to these activities was estimated at US$253, lower than the average facility-based delivery cost of US$331, indicating high cost-effectiveness51. However, when program management costs were included in the analysis, the broader estimate rose to US$2013 per delivery.
Notably, studies that focused on Indigenous or marginalised populations emphasised the value of cultural safety, continuity of care, and trusted relationships with health workers. However, some volunteer-based models (eg in Bolivia) faced sustainability challenges due to high fixed costs and limited reach, raising concerns about scalability30.
Clinical and diagnostic innovations
Clinical or diagnostic interventions such as misoprostol distribution, rapid syphilis screening, and neonatal resuscitation training offered cost-effective solutions for critical periods in maternal and newborn care37,44,55,57,60-63. In India, a modelling study estimated 7.5 maternal deaths averted (ICER of US$2001 per death averted) by administering misoprostol after delivery, compared to no intervention60. The 7.5 deaths averted occurred over a single delivery episode (per 10,000 home births), with a time horizon of approximately 2 days postpartum. A related study found that administering misoprostol for treatment versus prevention was highly cost-effective, with ICERs of US$8.52 per DALY averted61. Similarly, in Senegal, a trial comparing misoprostol and oxytocin against standard care found both strategies to be cost-effective for preventing severe postpartum haemorrhage, with ICERs of US$53 and US$159, respectively63. South Africa’s rapid syphilis screening using immunochromatographic strip testing averted 82% of congenital syphilis cases at an ICER of US$173 per case averted37. In addition, an economic model from Haiti found that integrating rapid syphilis tests into existing prenatal HIV screening programs was effective, with DALY costs of US$10.86 in rural areas57. Training traditional birth attendants in Zambia in neonatal resuscitation and infection management reported a 45% reduction in neonatal mortality, with ICERs ranging from US$36 to US$179 per DALY and US$2790 per neonatal death averted55.
In a nutritional intervention in Bangladesh, an early maternal micronutrient supplementation at 9 weeks of pregnancy had achieved under-five mortality and stunting reductions at a cost of US$30.92 per DALY averted, compared to standard care of 60 mg iron and 400 μg folic acid at 20 weeks62. Meanwhile, a model-based evaluation in India found that integrated reproductive health services, including family planning, safe abortion, and stepped-up emergency obstetric care could prevent 150,000 maternal deaths, with ICERs ranging from US$224.31 to US$448.62 per life year saved44.
Health system strengthening
Health system strengthening interventions targeted various components of the health system, including workforce development, infrastructure upgrades, supervision and mentorship, referral systems, and integration of services across care levels31,33,39,41,42,46,48,51,56,59,66. Several interventions demonstrated strong cost-effectiveness. The Ethiopian Millennium Rural Initiative improved the infrastructure, staffing, and referral systems of 30 primary healthcare units was highly cost-effective, with a cost per life saved of US$696842. Emergency obstetric ambulance service in Ethiopia and Uganda had remarkably low costs of US$32.14 and US$22.19 per life year saved, respectively33,59. Rwanda’s Mentorship, Enhanced Supervision for Healthcare and Quality Improvement program improved antenatal care quality at a cost of US$3.30 per complete clinical assessment48. Similarly, Burkina Faso’s Skilled Care Initiative, combining facility upgrades with behavioural change strategies, demonstrated incremental cost per delivery between US$253 and US$2013, depending on cost inclusions51.
Tanzania’s implementation of electronic clinical decision-support system within antenatal care and childbirth services improved interpersonal care and history taking, but it yielded limited health gains. The ICERs were high at US$3213 per 1% ANC quality improvement and US$440 per 1% improvement in childbirth care56.
A study in Malawi evaluated the combination of community mobilisation, facility quality improvement, and both together. Individually, the community component was more cost-effective (at US$104.73 per DALY averted) than the facility-only arm, while the combined arm produced the greatest overall health benefit, with an ICER of USD$194 per DALY41. These findings highlight the added value of integrating community demand generation with facility quality improvements.
Digital and mobile health tools
Digital health interventions, particularly mobile-based tools, also showed promising economic returns. These interventions were primarily designed to support care-seeking behaviour, improve quality of care, and strengthen the efficiency of CHW programs and health facilities. Interventions included text or voice message reminders to pregnant and postpartum women26,28, digital job aids for frontline health workers27,28,50, and electronic clinical decision-support systems integrated into antenatal and childbirth services56.
Bangladesh’s mCARE program, which used text-message reminders and CHW home visits, reported a cost per DALY averted of just US$40, and US$1149 per newborn death averted26. Similarly, the ReMiND program in India, which equipped accredited social health activists with mobile job aids to support maternal and newborn counselling and follow-up, was cost-saving from the societal perspective and cost-effective from the health system perspective, with an ICER of US$116 per DALY averted27. These digital interventions demonstrated significant impact through improved preventive care uptake and strengthened referral systems.
Other interventions included large-scale platforms like MOTECH in Ghana, which combined voice messaging and digital records with a cost of only US$27 per DALY averted28. The ImTeCHO platform in India, which provided multimedia content, task reminders, and decision-support tools to accredited social health activists in tribal and rural communities, reported a cost per life year saved of US$94 and was considered highly cost-effective50.
Results were modest for the Tanzania QUALMAT project, where electronic clinical decision-support system improved some quality in ANC and childbirth care but yielded high ICERs per 1% quality gain (US$3273 for ANC and US$448 for childbirth)56. These findings suggest that while digital tools can improve service quality, their effectiveness depends on local capacity, infrastructure, and health worker engagement.
Financial incentives
Financial incentive interventions such as conditional cash transfers, vouchers, performance-based financing, and non-monetary incentives such as delivery kits were effective when well implemented and aligned with supply-side capacity24,40,53,64,65.
In Uganda, a combined voucher and quality improvement program led to a 52% increase in institutional deliveries and had an ICER of US$424 per DALY averted24. A results-based financing program in Zambia was also cost-effective, with ICERs ranging from US$1072 to US$1324 per QALY depending on quality adjustments65. The Malawi Results-Based Financing for Maternal and Neonatal Health program, combining conditional cash transfers and infrastructure upgrades, achieved health gains but was sensitive to maternal life expectancy and quality assumptions. The ICER was reported as US$1487 per DALY averted and US$34760 per death averted, placing it in the cost-effective range at higher willingness-to-pay thresholds40. In contrast, Kenya’s Afya trial, which offered mobile cash transfers of US$9.32 per ANC, delivery, and postnatal visit attended had limited impact and high ICERs (US$1227 per additional ANC visit), mainly due to implementation delays and low transfer amounts53. Non-monetary incentive models such as one in Zambia that provides ‘mama’ kits (consisting of items like diapers, baby wraps, and blankets) increased facility deliveries, but with a higher ICER of US$6871 per death averted64.
Discussion
The findings of this review offer important insights for policymakers seeking to improve maternal health outcomes in Indigenous and rural populations. At the national level, the evidence can guide ministries of health and Indigenous health authorities in developing equitable funding frameworks and integrating culturally responsive care into universal coverage strategies. At the regional and district levels, the results can assist maternal health managers in prioritising cost-effective models that improve access and outcomes in underserved areas. These findings are also relevant to non-government organisations, philanthropic foundations, and research funders seeking high-impact interventions for Indigenous and rural populations. Interventions that were consistently cost-effective shared common features: they were contextually adapted, community-embedded, and integrated within broader health systems. These characteristics should serve as key guiding principles when designing or scaling programs across different implementation levels. Community-based and culturally tailored care models should be prioritised in regions with high maternal health disparities, especially where trust in formal health services is low67. These approaches are actionable for district health offices, community health managers, and non-governmental partners who coordinate grassroots maternal programs. Programs that engaged local actors such as CHWs, midwives, or traditional leaders improved service uptake and maternal and newborn outcomes68. For clinicians and midwifery leaders, these findings highlight the importance of maintaining continuity of culturally respectful care. Policy and donor agencies can use this evidence to justify investment in CHW and midwifery capacity-building. Where successful examples already exist, adapting these models for other Indigenous or rural populations with similar sociocultural dynamics could enhance health equity while maintaining cost-efficiency.
Digital health tools represent another area for strategic investment, especially in settings with expanding mobile coverage. These innovations are relevant to health system managers, digital health units within ministries, and philanthropic funders supporting technology-enabled care. However, their deployment should be accompanied by adequate training, human support, and system-level coordination to ensure sustainability. Standalone digital tools are unlikely to succeed unless embedded in functional care systems69,70. For clinicians and CHWs, mobile health platforms can improve efficiency and follow-up, while national digital health strategies can use this evidence to strengthen interoperability and accountability frameworks. Policymakers should therefore view mobile health as a complement to human resources rather than a substitute.
Financial incentive programs also merit close consideration, particularly by national financing agencies, development partners, and local implementers designing conditional cash transfers or performance-based schemes71. These mechanisms have shown promise but require careful design. Targeting mechanisms, transfer amounts, and alignment with quality improvements all influenced cost-effectiveness72. To maximise value, financial incentives should be integrated into quality improvement frameworks that involve both health facility readiness and community uptake, ensuring alignment between national funding priorities and local service delivery capacity.
Beyond community-based approaches, the cost-effectiveness of interventions varied across countries according to health-system structure, financing mechanisms, and geographic context. In high-income, colonisation settings such as Australia and Canada, efficiency gains were often achieved through culturally governed Indigenous health services integrated within publicly funded systems11,73. Conversely, in low- and middle-income countries, cost-effectiveness frequently resulted from improvements in basic system components such as ambulance networks, referral pathways, and workforce capacity. For district-level policymakers, these findings reinforce the economic justification for continued investment in basic system capacity. Geographic remoteness also shaped outcomes, as higher implementation costs in remote regions were often offset by improvements in access, continuity of care, and maternal health equity, a key consideration for rural planning divisions and transport-focused donors.
Across Indigenous and rural settings, several other public health interventions have also demonstrated cost-effective outcomes. These include prevention and management strategies for non-communicable diseases such as hypertension and diabetic retinopathy, as well as infectious disease interventions like screening and treatment of hepatitis b and respiratory syncytial virus prophylaxis among high-risk infants74-76. Preventive programs have likewise shown cost-effective outcome, including school-based physical activity and nutrition initiatives, road-injury prevention measures, and community-based drowning and suicide prevention programs77,78. These examples provide a useful backdrop for interpreting the economic value of maternal health interventions, as maternal health outcomes serve as a critical determinant of long-term community wellbeing and health system performance.
The time span of the included studies (2004–2025) introduces temporal heterogeneity in both maternal health systems and outcomes. Over this period, global maternal mortality declined, health financing models evolved from donor-dependent and fragmented funding structures to more integrated, performance-based, and government-financed systems, and new technologies such as mobile health platforms and point-of-care diagnostics emerged79. At the same time, economic evaluations evolved from narrowly scoped analyses with limited costing and shorter time horizons to more comprehensive assessments incorporating system-level costs, broader outcome measures, and real-world implementation constraints. These shifts influenced both intervention costs and effectiveness, explaining some variation in reported ICERs across decades. As a result, cost-effectiveness should not be viewed as a fixed attribute of interventions, but as contingent on evolving health-system contexts and methodological practices. This temporal perspective is relevant for researchers, donors, and policy agencies evaluating long-term trends in the value of maternal health interventions.
Several limitations of this review should be considered when interpreting the findings. First, the inclusion criteria focused strictly on full economic evaluations that compared both costs and health outcomes. As a result, the review excluded partial evaluations, return-on-investment analyses, and budget impact studies. While this approach enhanced methodological consistency, it may have omitted relevant interventions, particularly those implemented by non-government or community-based organisations operating outside formal health systems.
Second, the evidence base was heavily weighted toward studies conducted in low- and middle-income countries. There was limited representation of economic evaluations from high-income settings with Indigenous populations, particularly outside of Australia. This uneven distribution restricts the applicability of findings to contexts with very different health system structures, financing arrangements, or service delivery models.
Third, considerable variation existed in intervention types, costing methods, time horizons, and outcome measures across studies. These differences limited the feasibility of quantitative synthesis and introduced challenges in comparing cost-effectiveness across contexts. A narrative approach was used to manage this heterogeneity, but underlying inconsistencies in analytic methods, perspectives, and outcome valuation may still affect comparability.
Fourth, although the methodological quality of studies was generally acceptable based on the CHEC-Extended checklist and ECOBIAS appraisal, many evaluations lacked key components such as long-term follow-up, broad costing perspectives, or detailed sensitivity analyses. A few studies relied on intermediate outcomes like increased ANC visits rather than final health outcomes such as maternal mortality or DALYs, which weakens confidence in the strength of economic conclusions.
Finally, no weighting was applied to study quality during synthesis. While studies with high risk of bias were noted, all findings were discussed equally, potentially introducing variation in evidence strength across intervention categories.
This review identified key gaps in the economic evidence for maternal health interventions targeting Indigenous and rural populations.
First, while many evaluations focused on antenatal care and delivery, few examined the postpartum period. This evidence gap should prompt research funders and program designers to prioritise evaluation of mental health, breastfeeding, or long-term maternal wellbeing.
Second, most studies adopted narrow perspectives, typically those of the provider or health system, with limited attention to broader societal costs or benefits. For health economists and policymakers, expanding to societal perspectives, including indirect costs such as transportation, productivity loss, and informal caregiving, would more accurately capture the true value of maternal programs.
Third, studies often used short time frames and lacked robust uncertainty analysis. Researchers and academic collaborators should therefore design future economic evaluations with longer follow-up and thorough sensitivity testing to assess sustainability.
Finally, there is a need for high-quality economic evaluations in high-income countries with Indigenous populations where persistent disparities demand context-specific, culturally appropriate solutions, an agenda relevant to national health research councils, Indigenous-led organisations, and international funding agencies.
Conclusion
This systematic review aimed to evaluate the economic effectiveness of maternal healthcare interventions targeting Indigenous and rural populations. In doing so, it addressed a critical gap in understanding how health resources can be allocated efficiently in settings marked by structural and access-related barriers.
The review found that community-based, culturally adapted, and integrated service models were most consistently associated with favourable cost-effectiveness outcomes. Interventions that aligned with local needs and delivery systems tended to achieve better health outcomes at lower or comparable costs, particularly in resource-limited settings. However, the evidence base remains uneven, with limited evaluations from high-income Indigenous contexts and underrepresentation of interventions beyond antenatal and delivery care.
These findings offer a useful evidence base for policymakers seeking to improve maternal health outcomes in underserved populations while ensuring efficient use of limited resources. Integrating economic evaluations into maternal health program planning, especially for culturally adapted and community-delivered models, can support more informed, equitable health investment decisions.
Further research that addresses current gaps, adopts broader perspectives, and reflects local implementation realities will be essential to advancing economic evidence for maternal health interventions in diverse global settings.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflicts of interest
The authors declare no conflicts of interest.
AI disclosure statement
The authors used ChatGPT (OpenAI, GPT-4o version) in editing the manuscript. The authors verified accuracy of all AI-generated changes and take full responsibility for the final published work.



