Introduction
Cervical cancer is the third most common cancer in Nigeria, after prostate and breast cancer. According to Global Burden of Cancer (GLOBOCAN) 2022 estimates, 13,676 new cases and 7,093 deaths occurred in the country, with more than 28,320 women currently living with the disease1. Cancer survival in Sub-Saharan Africa remains poor, with nearly half of women diagnosed with cervical cancer likely to die within 3 years2. Because the majority of those affected are aged between 15 and 44 years (the reproductive years) the disease carries profound social, economic, and psychological consequences for women, their families, and their communities.
The high burden of cervical cancer in Nigeria is particularly worrisome given that the disease is largely preventable and curable if detected early3. Unfortunately, most cases present at advanced stages in health facilities, when little can be done to save the woman’s life4. While inadequate access to appropriate treatment facilities plays a role, late presentation has also been consistently linked to misconceptions, poor awareness, negative attitudes towards preventive practices, and suboptimal utilization of available screening and treatment options5,6.
In spite of national and global efforts to promote cervical cancer prevention, screening coverage in Nigeria remains low, particularly among women in the local community. This undermines progress towards WHO’s cervical cancer elimination targets and sustains preventable morbidity and mortality. Understanding how specific knowledge domains, attitudes, and contextual factors influence screening uptake among community-based women is therefore critical for designing effective, population-level interventions. Although several studies on women’s knowledge, attitudes, and practices regarding cervical cancer have been conducted in Nigeria, most have been based in hospitals or institutions7-10. Many of these studies did not examine the different domains of knowledge in detail. Furthermore, while some Nigerian studies have reported factors influencing knowledge, attitudes, and practices, their analyses were often unadjusted, leaving room for potential confounding. Even where adjusted analyses were performed, the studies typically had small sample sizes11-13. The present study therefore sought to address these gaps by assessing cervical cancer knowledge domains, attitude, and screening uptake among community-based women, and by identifying significant predictors of screening uptake, knowledge and attitudes.
Methods
Study design and setting
This cross-sectional study was conducted among community women in Omala local government area (LGA) of Kogi State, located in the North Central region of Nigeria. The area had an estimated population of 145,500 in 2022, of which approximately 49.6% were female14.
The inhabitants predominantly depend on agriculture, including farming and fishing, as their main sources of livelihood.
Sample size and recruitment of participants
The minimum required sample size was calculated using Cochran’s formula for single proportions: n = (Z² × p × (1 – p)) ÷ d² where Z=1.96 at 95% confidence level, p=0.435 (knowledge prevalence from a previous study in North Central Nigeria)15, and d=0.05 margin of error. This yielded a minimum sample size of 379. To account for a 12% anticipated non-response rate, the final sample size was adjusted to 425 participants.
Included in the study were women aged ≥15 years residing in the selected communities for at least 6 months and willing to participate. Women who were critically ill, non-residents, and those not at home during the period of the interview were excluded. Eligible women aged 15 years and above were recruited using a multistage sampling technique. Omala LGA was selected because it is predominantly rural, has limited access to organized cervical cancer screening services, and is representative of similar settings in North Central Nigeria. In the first stage, the LGA was stratified into four geographic zones (north, south, east, and west) to ensure spatial representativeness. The most populous community in each zone was purposively selected to maximize coverage and feasibility.
In the second stage, the sample size allocated to each community was determined proportionate to the female population of the zone (Supplementary table 1). Accordingly, 241 participants were recruited from Abejukolo (north), 83 from Bagana (west), 52 from Bagaji (south), and 49 from Ibado Akpacha (east). In the final stage, households were selected systematically within each community, and one eligible woman was randomly selected per household for interview.
Due to the absence of proper documentation of households and streets, systematic household sampling was applied. In Abejukolo, where houses are densely clustered, the community was subdivided into four sections, and one in every five households was selected until the required sample size was achieved.
In the remaining three communities, where household settlements were more dispersed, an average of one in three households on either side of the streets was sampled. Households were selected until the required sample size for each zone was achieved. In households with more than one eligible woman, priority was given to interviewing the mother and the first eligible daughter.
Data collection
The data collection instrument was a semi-structured questionnaire developed specifically for this study, drawing on items adapted from previously validated instruments used in similar settings. Content validity was ensured through expert review by public health specialists and epidemiologists with experience in cervical cancer research, who assessed the relevance, clarity, and cultural appropriateness of the items.
Interviews were conducted by trained female research assistants with at least a diploma certificate. They underwent a day training on study objectives, ethical conduct, administration of the questionnaire, translation into the local language, and a mock interview session. Participants were recruited through household visits in the selected communities by trained female research assistants who were familiar with the local context. Eligible women were approached in their homes, the purpose of the study was explained in detail, and verbal informed consent was obtained prior to participation. Literate participants completed the questionnaire independently, while the trained interviewers administered the questionnaire to participants with limited literacy using a face-to-face interview format. Interviewers provided clarification where necessary without influencing responses. Participants were allowed sufficient time to complete the questionnaire at their own pace, with completion taking 20–30 minutes, on average. Most questionnaires were retrieved on the spot, while a few were collected during a second visit at the participant’s request. Data collection was conducted between September and October 2023.
Measurement of variables and statistical analysis
Level of knowledge
Four domains of knowledge were assessed: general knowledge (two items), knowledge of cervical cancer risk factors (13 items), knowledge of signs and symptoms (six items), and knowledge of screening (three items). Each item was assessed using three response options: ‘yes’, ‘no’, and ‘I don’t know’. For scoring, a correct response (‘yes’) was assigned a score of 1, while ‘no’ or ‘I don’t know’ were assigned a score of 0. The knowledge score for each domain was calculated as the mean of all item scores within that domain, ranging from 0 (minimum) to 1 (maximum). Scores were interpreted as follows: 0 = little or no knowledge, 0.1–0.5 = poor knowledge, 0.6–1.0 = fair/good knowledge. A consolidated knowledge score was computed as the mean of the four domain-specific scores to provide an overall measure of cervical cancer knowledge.
Attitude
Attitude was assessed using a five-point Likert scale, ranging from ‘strongly disagree’ (1 point) to ‘strongly agree’ (5 points). For each participant, the attitude score was calculated as the average of all item scores, with possible values ranging from 1 (minimum) to 5 (maximum). An average score above 2.5 was interpreted as indicating a positive perception/attitude. To aid comparability, average scores were also standardized into percentages by dividing the mean score by the maximum score (5) and multiplying by 100.
Willingness to undergo screening (behavioural intention)
Willingness to undergo screening was assessed using three response options: ‘yes’, ‘no’, and ‘not sure’.
Uptake of cervical cancer screening
Uptake of cervical cancer screening was measured using a simple dichotomous response (‘yes’ or ‘no’) to the question, ‘Have you ever had a cervical cancer screening test (Pap test) before?’ The proportion of participants who responded ‘yes’ was expressed as a percentage to represent the level of screening uptake in the study population.
Statistical analysis
The distribution of study variables was summarized using frequencies and percentages, presented in tables. Multivariable logistic regression analysis was employed to identify independent predictors of good knowledge, positive attitudes, and cervical cancer screening uptake, while adjusting for potential covariates. Odds ratios (ORs) with 95% confidence intervals (CIs) were reported, with an OR greater than 1 indicating a positive association between the predictor and the outcome variable. Statistical significance was set at p<0.05. All analyses were conducted using Statistical Package for the Social Sciences v23 (IBM Corp; https://www.ibm.com/products/spss-statistics).
Ethics approval
The study was reviewed and approved by the National Open University of Nigeria Research Ethics Committee (ETC/2024/04/NOU214057599) . In addition, a written approval was obtained from the Department of Public Health Omala LGA, Kogi State Nigeria, before the commencement of the study. Oral informed consent was obtained from participants due to varying levels of literacy and because most participants were more comfortable with oral consent than written consent. This consent procedure was also approved by the ethics committee.
Results
A total of 416 questionnaires were completed out of the 425 distributed, representing a response rate of 97.9%. The sociodemographic characteristics of the participants are presented in Table 1. Most of the women were aged 40 years or less (71.7%). A majority were married (58.2%), had attained more than secondary education (53.0%), and were employed in either private or public institutions (31.7%).
Table 2 shows the distribution of knowledge across the four domains. Overall, 41.7% of participants demonstrated a consolidated knowledge score classified as fair/good. Across the domains, fair/good knowledge was highest for general knowledge (59.2%) and lowest for knowledge of cervical cancer risk factors (15.5%). Knowledge of signs and symptoms was 26.8%, while knowledge of cervical cancer screening was 45.2%.
With respect to specific items, only 42.3% of the participants correctly identified HPV infection as a risk factor. Although 64.6% recognized vaginal discharge as a symptom, much fewer identified vaginal bleeding after sexual intercourse (29.8%) and intermenstrual vaginal bleeding (18.5%) as symptoms. Furthermore, knowledge of appropriate screening intervals was particularly low, with only 16.6% correctly aware of how often screening should be done.
As shown in Figure 1, the most common source of information on cervical cancer was the mass media (36.7%), followed by hospitals (26.6%) and family/friends (21.9%).
Regarding attitudes (Supplementary table 2), most participants expressed generally positive attitudes toward cervical cancer prevention and control. The median attitude score was 3.3. Specifically, participants had high average scores for beliefs about the curability of cervical cancer when detected early (4.02), the effectiveness of vaccination for young girls (3.75), the limited role of charms and evil spirits (3.74), and the effectiveness of Pap smear screening (4.0). However, despite these favourable attitudes, only 36.0% of participants expressed willingness to undergo screening if adequately educated.
In terms of screening uptake (Table 3), only 12.2% of participants reported ever having had a cervical cancer test. The most commonly cited reasons for non-uptake were lack of knowledge about the test (23.3%), absence of symptoms (22.4%), and the belief that they were not personally at risk of cervical cancer (11.1%).
Multivariable logistic regression analysis identified several significant predictors of cervical cancer knowledge, attitudes, and screening uptake (Tables 4 and 5; Supplementary table 3).
Increasing educational attainment was significantly associated with good knowledge (p (trend)=0.001). Other factors positively associated with good knowledge included having fewer children (OR=2.62, 95%CI 1.34–5.11) compared to four or more children, a family history of cervical cancer (OR=3.72, 95%CI 1.18–11.68), Islamic compared to Christian faith (OR=2.13, 95%CI 1.26–3.57), increasing age (OR=1.07, 95%CI 1.03–1.11), and older age at first sexual intercourse (OR=1.04, 95%CI 1.00–1.08).
Positive attitudes were associated with higher educational attainment (p=0.037), nulliparity (OR=2.61, 95%CI 1.04–6.54), and having fewer children (OR=2.16, 95%CI 1.14–4.10) compared to four or more children (Supplementary table 3).
Higher attitude scores were associated with increased likelihood of screening uptake (p=0.006). Marital status also influenced uptake, with married women (OR=3.50, 95%CI 1.08–11.39) and divorced/separated women (OR=6.03, 95%CI 1.21–30.02) more likely to undergo screening compared to single women. Additionally, knowledge of cervical cancer screening showed a positive but borderline association with uptake (OR=2.54, 95%CI 1.00–6.69).
Table 1: Sociodemographic characteristics of rural women in Omala local government area, Kogi State, Nigeria
| Characteristic | Variable | n (%) |
|---|---|---|
| Age (years) (mean 34.7±10.9) | ≤30 | 160 (38.5) |
| 31–40 |
138 (33.2) |
|
| ≥41 |
113 (27.2) |
|
| Not supplied |
5 (1.2) |
|
| Religion | Islam | 197 (47.4) |
| Christianity |
210 (50.5) |
|
| Other |
4 (1) |
|
| Not supplied |
5 (1.2) |
|
| Marital status | Single | 117 (28.1) |
| Married |
242 (58.2) |
|
| Divorced |
28 (6.7) |
|
| Widow |
26 (6.3) |
|
| Not supplied |
3 (0.7) |
|
| Level of education | Informal | 43 (10.3) |
| Primary school |
32 (7.7) |
|
| Secondary school |
103 (24.8) |
|
| Tertiary education |
230 (53.0) |
|
| Not supplied |
8 (1.9) |
|
| Occupational status | Unemployed/home duties | 85 (20.4) |
| Self-employed |
118 (28.4) |
|
| Private organization |
74 (17.8) |
|
| Public service |
58 (13.9) |
|
| Not supplied |
81 (19.5) |
|
| Family history of cervical cancer | No | 383 (92.1) |
| Yes |
23 (5.5) |
|
| Not supplied |
10 (2.4) |
|
| Age at first birth (years) | 16–20 | 43 (10.3) |
| 21–25 |
132 (31.7) |
|
| 26–30 |
74 (17.4) |
|
| >30 |
21 (5) |
|
| Not supplied |
69 (16.6) |
|
| Number of pregnancies | 0 | 138 (33.2) |
| 1–3 |
138 (33.4 |
|
| 4–6 |
118 (28.4) |
|
| >6 |
18 (4.3) |
|
| Not supplied |
4 (1) |
|
| Age at first sexual intercourse (years) | 11–15 | 73 (17.5) |
| 16–20 |
164 (39.4) |
|
| 21–25 |
108 (26) |
|
| ≥26 |
34 (8.2) |
|
| Not supplied |
37 (8.9) |
|
| Mean |
18.1±7.6 |
Table 2: Distribution of knowledge about cervical cancer across four domains: general knowledge, risk factors, signs/symptoms, and screening
| Knowledge domain | Item/score | Frequency (%) |
|---|---|---|
| General knowledge | Ever heard about cervical cancer | 263/412 (63.8) |
| Awareness of how common cervical cancer is |
250/409 (61.1) |
|
| Mean score | 0 | 138 (33.7) |
| 0.1–0.5 |
29 (7.1) |
|
| 1 |
242 (59.2) |
|
| Knowledge of cervical cancer risk factors | HPV infection | 174/411 (42.3) |
| Chlamydia infection |
103/409 (25.2) |
|
| Having sex at age less than 18 years |
113/408 (27.7) |
|
| Having multiple sexual partners |
178/411 (43.3) |
|
| Having one partner who is having sex with other women |
178/411 (43.3) |
|
| Giving birth to many children |
58/408 (14.2) |
|
| First birth at less than age 20 years |
61/408 (15.0) |
|
| Smoking |
75/410 (18.3) |
|
| Low intake of fruits and vegetables |
61/408 (15.0) |
|
| HIV or compromised immune system |
212/411 (51.6) |
|
| Having a mother or sister who has had cervical cancer |
43/410 (10.5) |
|
| Being poor |
29/409 (7.1) |
|
| Using birth control pills for a long time |
108/409 (26.4) |
|
| Mean score | 0 | 62 (15) |
| 0.1–0.5 |
287 (69.5) |
|
| 1 |
64 (15.5) |
|
| Knowledge of cervical cancer signs /symptoms | Vaginal bleeding after sex | 119/399 (29.8) |
| Vaginal bleeding after menopause |
183/399 (45.9) |
|
| Vaginal bleeding between periods |
74/401 (18.5) |
|
| Vaginal discharge that is watery and has a strong odour or that contains blood |
259/401 (64.6) |
|
| Pelvic pain or pain during sex |
95/395 (24.1) |
|
| Painful urination or blood in the urine |
265/399 (66.4) |
|
| Mean score | 0 | 105 (25.6) |
| 0.1–0.5 |
195 (47.6) |
|
| 1 |
110 (26.8) |
|
| Knowledge of cervical cancer screening | Age for initiation of screening (Pap smear) | 227/380 (59.7) |
| Ever heard or read about cervical cancer screening |
232/379 (61.2) |
|
| Screening interval for one%u2019s age group |
63/379 (16.6) |
|
| Mean score | 0 | 65 (17) |
| 0.1–0.5 |
145 (37.8) |
|
| 1 |
173 (45.2) |
|
| Consolidated (overall) knowledge mean score | 0 | 2 (7.6) |
| 0.1–0.5 |
210 (50.7) |
|
| 1 |
171 (41.7) |
Table 3: Uptake of cervical cancer screening and reasons for non-utilization among rural women
| Characteristic | Variable | n (%) |
|---|---|---|
| Ever done HPV or Pap smear before (N=384) | No | 337 (87.8) |
| Yes |
47 (12.2) |
|
| Reasons for not going for screening test (N=416) | I do not know much about how it is done | 97 (23.3) |
| I do not know where to do it |
40 (9.6) |
|
| I do not want to get a bad result |
40 (9.6) |
|
| I have not had any sign or symptoms |
93 (22.4) |
|
| I believe I can never have cervical cancer |
46 (11.1) |
|
| The test is expensive |
29(7.0) |
|
| The test is embarrassing |
23 (5.5) |
|
| I do not want to be examined by a male doctor |
16 (3.8) |
|
| My partner or husband will not approve it |
20 (4.8) |
|
| The test is painful |
13 (3.1) |
Table 4: Predictors of good knowledge of cervical cancer among rural women, based on multivariable logistic regression
| Characteristic | Variable | Poor knowledge n (%) or mean±SD | Good knowledge n (%) or mean±SD | OR (95%CI) | p-value |
|---|---|---|---|---|---|
| Educational attainment | Primary or less | 60 (25.2) | 15 (8.8) | 1.00 (ref) | |
| Secondary |
62 (26.1) |
41 (24.1) | 4.05 (1.54–10.6)† | ||
| Tertiary |
116 (48.7) |
114 (67.1) | 5.96 (2.33–15.2)† | 0.001** | |
| Occupational status | Unemployed/home duties | 55 (29.1) | 30 (20.5) | 0.96 (0.46–2.02) | |
| Self-employed |
59 (31.2) |
59 (40.4) | 1.59 (0.88–2.90) | ||
| Private /public worker |
75 (39.7) |
57 (39.0) | 1.00 (ref) | ||
| Marital status | Single | 55 (24.1) | 62 (34.3) | 2.68 (0.75–9.53) | |
| Married |
131 (57.5) |
107 (59.1) | 2.14 (0.85–5.39) | ||
| Divorced/widowed |
42 (18.4) |
12 (6.6) | 1.00 (ref) | ||
| Parity | Nulliparous | 81 (33.5) | 57 (33.5) | 1.96 (0.76–5.09) | |
| 1–3 pregnancies |
70 (28.9) |
68 (40.0) | 2.62 (1.34–5.11)† | ||
| ≥4 pregnancies |
91 (37.6) |
45 (26.5) | 1.00 (ref) | ||
| Family history of cervical cancer | No | 222 (93.7) | 161 (95.3) | 3.72 (1.18–11.68)† | |
| Yes |
15 (6.3) |
8 (4.7) | 1.00 (ref) | ||
| Religion | Islam | 103 (43.1) | 94 (56.0) | 2.13 (1.26–3.57)† | |
| Christianity |
136 (56.9) |
74 (44.0) | 1.00 (ref) | ||
| Age | Mean |
34.54±10.81 |
35.01±10.96 | 1.07 (1.03–1.11)† | |
| Age at first sexual intercourse | Mean |
17.27±10.96 |
19.35±7.29 | 1.04 (1.00–1.08)† | 0.054 |
*p<0.05, **p<0.01, ***p<0.001.
† Statistically significant (95% confidence interval).
CI, confidence interval. OR, odds ratio. ref, reference. SD, standard deviation.
Table 5: Predictors of cervical cancer screening uptake among rural women, based on multivariable logistic regression
| Characteristic | Variable | Screened for cervical cancer | OR (95%CI) | p-value | |
|---|---|---|---|---|---|
|
No |
Yes |
||||
| Educational attainment | Primary school or less | 65 (19.5) | 8 (17.0) | 1.00 (ref) | |
| Secondary school |
89 (26.7) |
10 (21.3) | 0.73 (0.24–2.24) | ||
| Tertiary education |
179 (53.8) |
29 (61.7) | 0.72 (0.26–2.00) | ||
| Marital status | Single | 107 (30.3) | 7 (14.9) | 1.00 (ref) | |
| Married |
192 (57.0) |
33 (70.2) | 3.50 (1.08–11.39)† | ||
| Divorced/widowed |
43 (12.8) |
7 (14.9) | 6.03 (1.21–30.02)† | ||
| Family history of cervical cancer | No | 312 (94.3) | 42 (91.3) | 1.00 (ref) | |
| Yes |
19 (5.7) |
4 (8.7) | 0.97 (0.24–3.89) | ||
| Knowledge of cervical cancer screening | No | 137 (42.2) | 8 (17.0) | 1.00 (ref) | |
| Yes |
188 (57.8) |
39 (83.0) | 2.54 (1.00–6.69)† | ||
| Religion | Islam | 164 (49.2) | 21 (45.7) | 0.96 (0.49–1.89) | |
| Christianity |
169 (50.8) |
25 (54.3) | 1.00 (ref) | ||
| Age (mean±SD) | 36.40±11.91 | 34.00±10.19 | 0.98 (0.93–1.04) | ||
| Number of pregnancies (median±SD) | 3±2.42 | 2±2.07 | 1.04 (0.84–1.30) | ||
| Attitude score (mean±SD) | 3.10±0.40 | 3.50±0.54 | 2.85 (1.36–5.99)† | 0.006** | |
| Risk factor awareness score (mean±SD) | 0.17±0.18 | 0.36±0.21 | 0.73 (0.11–4.75) | ||
| Signs/symptom awareness score (mean±SD) | 0.29±0.32 | 0.48±0.26 | 0.82 (0.24–2.84) | ||
*p<0.05, **p<0.01, ***p<0.001.
† Statistically significant (95% confidence interval).
CI, confidence interval. OR, odds ratio. ref, reference. SD, standard deviation.
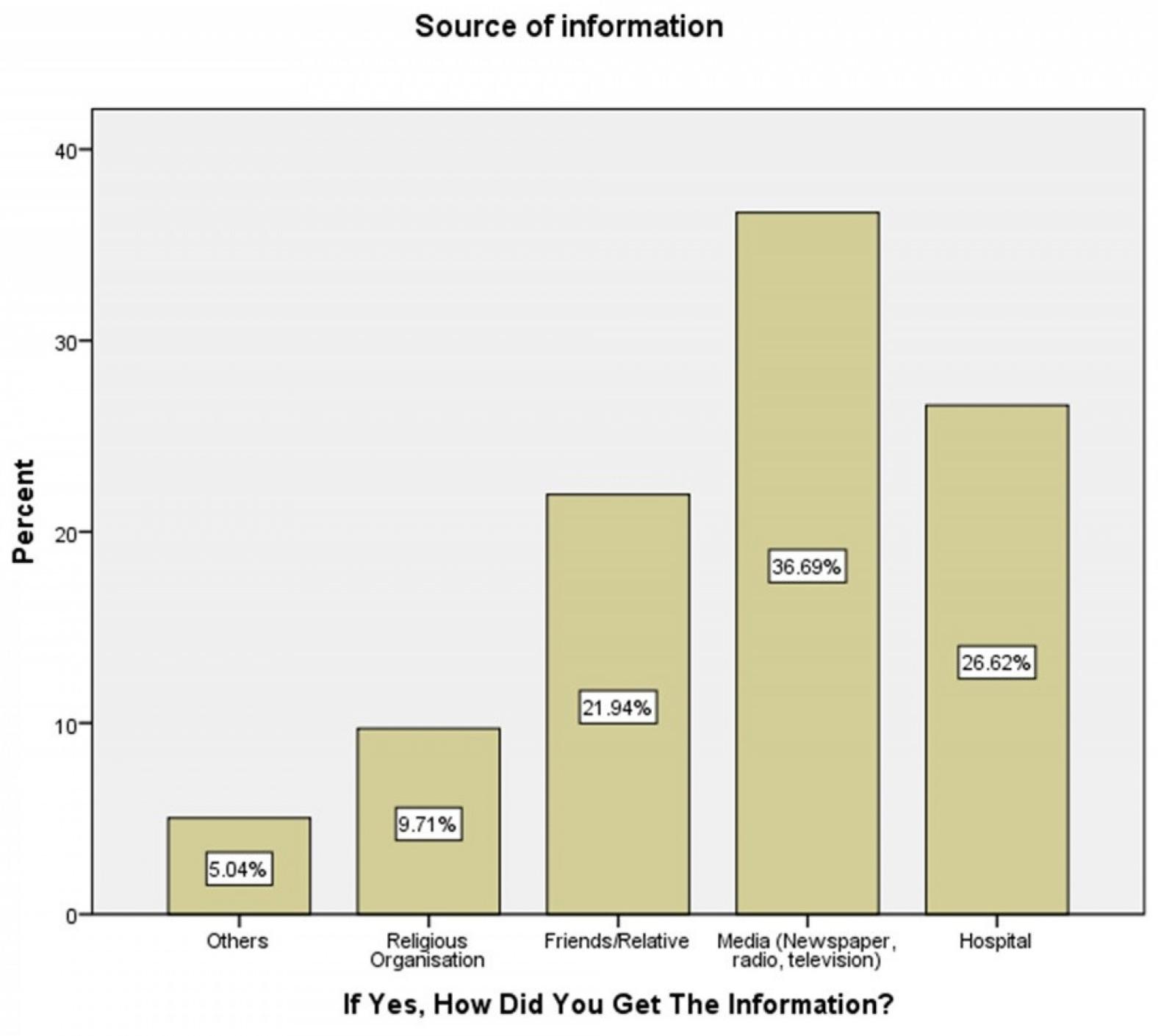 Figure 1: Sources of information about cervical cancer among rural women in Omala local government area, Nigeria.
Figure 1: Sources of information about cervical cancer among rural women in Omala local government area, Nigeria.
Discussion
The study revealed overall poor knowledge of cervical cancer among participants (41.7%), with the lowest scores observed for knowledge of risk factors (15.5%), followed by signs and symptoms (26.8%) and screening methods (45.2%). Mass media was the most common source of information (36.7%). Despite this, a majority of participants exhibited a positive attitude to cervical cancer (66%), although willingness to go for screening was low (36.6%). Uptake of cervical cancer screening was very low (12.2%), primarily due to lack of awareness about the test (23.3%). Significant predictors of good knowledge included higher educational attainment, lower parity, family history of cervical cancer, increasing age, and older age at first sexual intercourse. Key predictors of screening uptake were higher attitude scores, being married, and knowledge of cervical cancer screening.
The consolidated knowledge score in our study (41.7%) was higher than the 8.3% reported among rural women in Lagos, South West Nigeria15. Variations in scores across studies may reflect differences in the number of items included in the questionnaire and the way questions were framed. Notably, most existing studies did not assess or report an overall or consolidated knowledge score, which limits direct comparisons. We recommend that future studies include an evaluation of overall knowledge, as this provides a more comprehensive understanding of participants’ awareness. Nevertheless, our findings on general knowledge were comparable to previous reports, including 66.9% among market women in Sabon Gari, Zaria, Nigeria (2013) and 67% in Ilorin West, Nigeria (2015)12,14. Notably, although Zaria and Ilorin are urban areas, they are located within the same geopolitical zone as our study population, which may explain some similarities in findings. Our general knowledge score was higher than the 15–31.2% reported among rural women in the South West region of Nigeria11,15,16, suggesting that regional differences may play a role. Conversely, it was lower than scores observed among health workers and women attending tertiary hospitals in the South South and South West regions7,9, likely reflecting differences in educational background and access to health information. With respect to specific knowledge domains, the average score for cervical cancer risk factors in our study was consistent with a Lagos-based study9. However, knowledge of signs and symptoms was lower than observations in that study, which is understandable given that the population in Lagos was more educated. Higher recognition of painful urination and vaginal discharge as signs of cervical cancer in our study aligns with observations among market women in Zaria, North Central Nigeria14. In contrast, vaginal bleeding was more commonly recognized in several other studies8,9,11. Similarly, the higher recognition of multiple sexual partners as a risk factor in our study is consistent with findings from other regions9,11. Regarding knowledge of cervical cancer screening, the proportion of participants with good knowledge in our study was comparable to observations in Zaria and Ilorin12,15, but was higher than the observations in South West Nigeria11,16. These differences may reflect regional variations or differences in educational exposure. Finally, the most common source of information in our study, mass media, was consistent with findings from Ilorin and Lagos, but differed from those of Zaria and Ogbomoso, where hospitals and healthcare workers were reported as the predominant sources of knowledge11,15.
The overall positive attitudes toward cervical cancer treatment and prevention in our study aligns with findings from a study in South West Nigeria7. However, unlike that study – in which all respondents expressed willingness to undergo screening if given the opportunity – only 36% of participants in our study indicated willingness to be screened if adequately informed. This difference may reflect better health-seeking behaviour in the hospital-based population of the previous study. Other studies have reported poor attitudes scores and attitudes toward cervical cancer5,8. Cervical cancer screening uptake in our study was higher than that reported among community women in South West Nigeria, possibly due to differences in educational exposure11,16-18. Conversely, uptake was lower than observed in previous studies among urban women in North Central Nigeria12,15. Poor awareness was consistently reported as the main barrier to screening across studies19-21. Our findings that higher educational attainment, increasing maternal age, and low parity were predictors of cervical cancer knowledge are supported by previous Nigerian studies demonstrating that higher socioeconomic status is associated with better knowledge22,23. Notably, most existing studies in Nigeria did not assess predictors of knowledge, and those that did often lacked adjusted analyses23. Nonetheless, our results align with predictors of good knowledge and positive attitude reported in Saudi Arabia24. Regarding cervical cancer screening uptake, our findings are consistent with previous observations from similar populations12,13.
Our findings on knowledge highlight a critical gap in awareness creation. Effective prevention of cervical cancer requires reducing exposure to key risk factors, including HPV infection, risky sexual behaviours, and grand multiparity. Such preventive efforts are challenging in populations with low awareness. High incidence of cervical cancer and subtypes associated with family history have been previously reported in Nigeria25. The generally poor knowledge of signs and symptoms except for vaginal discharge suggests that many women in this population may present late for treatment if they develop cervical cancer. Timely presentation depends on the ability to recognize key warning signs. Although a majority of participants had heard of cervical cancer screening, only a small proportion knew the recommended frequency of testing, indicating that underutilization of available services is likely to persist, especially if initial screening results are negative. Despite overall positive attitudes, understanding was insufficient, as only 36% expressed willingness to undergo screening if adequately informed. This underscores the need for community-based awareness programs targeting specific domains of knowledge and attitudes, even among those with general awareness of the disease. The observed gap between knowledge and practice is concerning, suggesting that awareness alone may not translate into preventive action. Nevertheless, the positive association between specific knowledge of cervical cancer screening and screening uptake is encouraging, indicating that targeted education could improve utilization. This aligns with the finding that poor awareness was the main reason cited by participants for not undergoing screening.
The observed role of educational attainment and family history of cervical cancer as predictors of knowledge may reflect increased access to information among these groups. Similarly, the predictive effect of increasing age could be related to its association with aspects of socioeconomic status, such as employment, education, wealth, and social networks, which facilitate exposure to health information. The association with Islamic religion may reflect potential selection bias, as women of lower socioeconomic status in some Islamic communities may have been less likely to participate due to religiocultural restrictions or misconceptions. Likewise, the influence of higher educational attainment and low parity on positive attitude may indicate that women with higher socioeconomic status are generally more open and receptive to new health information26. Women with higher socioeconomic status are also less likely to hold superstitious beliefs compared to their counterparts. In our study, positive attitudes emerged as the strongest predictor of cervical cancer screening uptake, suggesting that the influence of educational attainment on uptake is largely mediated through its effect on attitudes. This insight should inform the development of targeted health education programs aimed at improving attitudes and ultimately reducing the burden of cervical cancer in the population.
The findings underscore the need for context-specific strategies in rural communities to improve cervical cancer prevention. Strengthening local healthcare infrastructure, including mobile screening units and integration of cervical cancer services into primary health care, could enhance accessibility. Engaging community leaders and using culturally appropriate health messaging may help overcome sociocultural barriers, while training and empowering local health workers to deliver education and screening services could ensure sustainability.
This study has some limitations that should be considered when interpreting the findings. First, its cross-sectional design means that predictors and outcomes were measured simultaneously, and therefore reverse causation cannot be ruled out. Second, as the data were based on self-reports, there is the possibility of information bias. Finally, the findings are context-specific and may not be generalizable beyond similar rural settings. the use of interviewer-administered questionnaires among a small proportion of uneducated participants could introduce interviewer bias. However, interviews were conducted to ensure comprehension and inclusion of participants who might otherwise have been excluded. To mitigate potential interviewer bias, standardized interviewer training, including adherence to wordings and translation in local language, was done to address potential bias. Future analytical or experimental studies, using appropriately selected controls, are recommended to confirm these associations and strengthen causal inferences.
Conclusion
The study revealed poor knowledge of cervical cancer among rural women in Omala LGA, with notable variations across knowledge domains. Uptake of cervical cancer screening was low, and while some aspects of attitudes were positive, willingness to undergo screening remained poor. These findings underscore the need to strengthen educational opportunities and improve the socioeconomic conditions of rural women, alongside implementing specifically designed awareness programs. Such interventions should aim to provide comprehensive knowledge of cervical cancer and address misconceptions, superstitious beliefs, fear, and negative attitudes that hinder screening uptake in rural settings.
Acknowledgements
We appreciate the department of public health, Omala LGA of Kogi State, for supporting the study. We also appreciate all community women who willingly participated in the study. We hereby acknowledge our interviewers who assisted in the data collection process.
Funding
The authors received no funding for this study.
Conflicts of interest
The authors declare no conflict of interest.
AI disclosure statement
ChatGPT was used to assist with minor grammatical corrections and improvements in clarity in selected statements in the abstract, methodology, and the final paragraph of the discussion. The scientific content, analysis, and conclusions remain entirely the authors’ own. All edits by ChatGPT were reviewed and the authors take full responsibility for the final article content.
References
Supplementary material is available on the live site https://www.rrh.org.au/journal/article/10433/#supplementary
